ACID MINE DRAINAGE
Acid mine drainage (AMD) is often a result from mining operations. Low-pH water is produced by the oxidation of sulfide (usually from the iron-sulfide mineral pyrite) into sulfuric acid. Often AMD-impacted springs and streams are easy to identify due to the bright orange color resulting from oxidation of Fe(II) into Fe(III), which precipitates as iron-oxides (essentially rust). Oxidation of pyrite is a natural process that occurs as weathering of rocks with pyrite slowly exposes the mineral to the atmosphere, allowing it to react with oxygen in a reaction that is often catalyzed by microorganisms. However, mining operations effectively accelerate this process by exposing pyrite-containing rocks to the atmosphere and oxygen-containing groundwater much faster than natural weathering rates.
|
AMD-impacted spring near the CO-OP coal mine in Kentucky. The emergence is near the base of the large boulder to the upper left. Note the orange color due to precipitation of iron oxides. Photo by Jeff Havig.
|
AMD-impacted spring emergence (lower center) mixing with a clear stream (top center) in the Wildcat Branch area of Kentucky, where historical strip mining for coal was conducted. While the iron in the water is easy to see due to the bright orange iron oxide precipitate, other toxic elements such as arsenic, mercury, cadmium and lead may also be present and not visible. Photo by Jeff Havig.
|
An interesting microbial community living in an AMD-impacted spring in Kentucky. Microorganisms that live in these springs may provide clues for how to remediate the effects of AMD on streams and rivers. Photo by Jeff Havig.
|
Low pH produced by the chemical oxidation of pyrite is detrimental to multicellular organisms as well as microorganisms that are not adapted to low pH, and other potentially dangerous elements are released (including chromium, nickel, copper, zinc, arsenic, cadmium, mercury, and lead) impacting affected soil, forests, wetlands, streams, rivers, and groundwater. A total of 26 states produce coal, with the top six including (in descending order) Wyoming, West Virginia, Kentucky, Pennsylvania, Texas, and Montana. In the US alone, over 9,300 miles of streams and rivers are impacted by AMD (Rimstidt and Vaughan, 2014). A legacy of coal mining operations has left impacts in every coal-producing state, including the Daniel Boone National Forest in south-central Kentucky. We are currently exploring AMD sites in this area, looking to characterize the geochemistry and microbiology of AMD-impacted springs in the area. We hope to contribute to solutions for mitigating the effects of AMD through building a better understanding of the impact of microbial communities on altering the geochemistry. Also, we are interested in learning about systems that have high Fe(II) and low dissolved oxygen and what they might teach us about life on the early earth before, during, and following the great oxidation event when the Earth's atmosphere switched from reducing to oxidizing around 2.5 billion years ago.
Below is a manuscript that we have published with our colleagues from work done at these sites:
Below is a manuscript that we have published with our colleagues from work done at these sites:
|
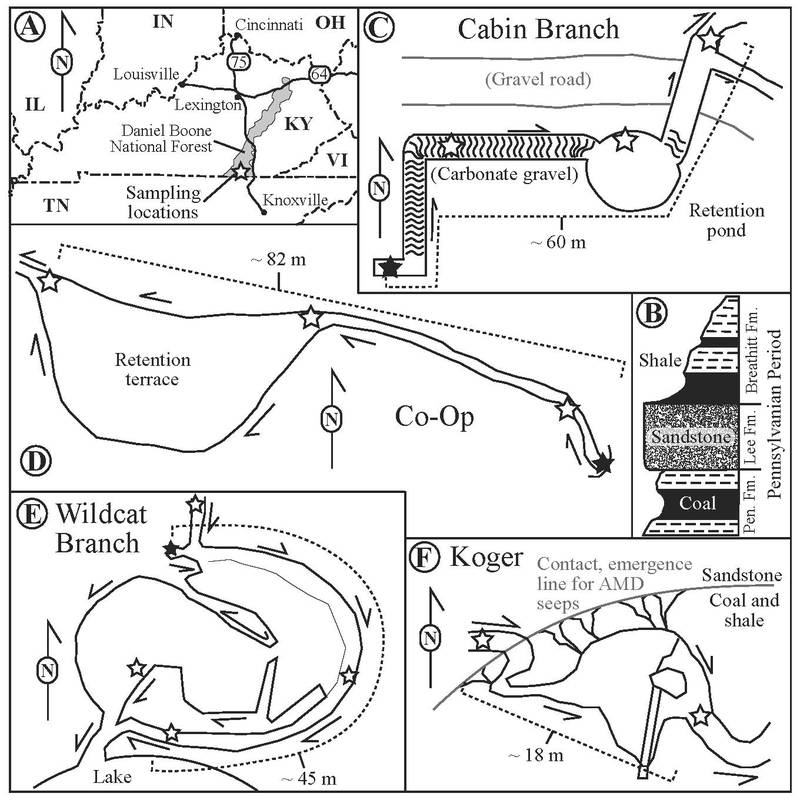
Havig et al. (2017) Figure 1: Maps and geology of the sampling area in the southern portion of the Daniel Boone National Forest, Kentucky, USA. A) Location Map. B) Idealized cross section representing the local geology, not to scale. C) Cabin Branch sampling area. D) Co-Operative Mine sampling area. E) Wildcat Branch sampling area (Note: YOU-glena and No-glena sites are located off map, approximately 250 m WSW from Wildcat sampling area). F) Koger Branch sampling area. Stars denote sampling locations, and filled stars denote emergences. Arrows indicate the direction of flow. Note that surface sketch maps are idealized and not to scale.
|
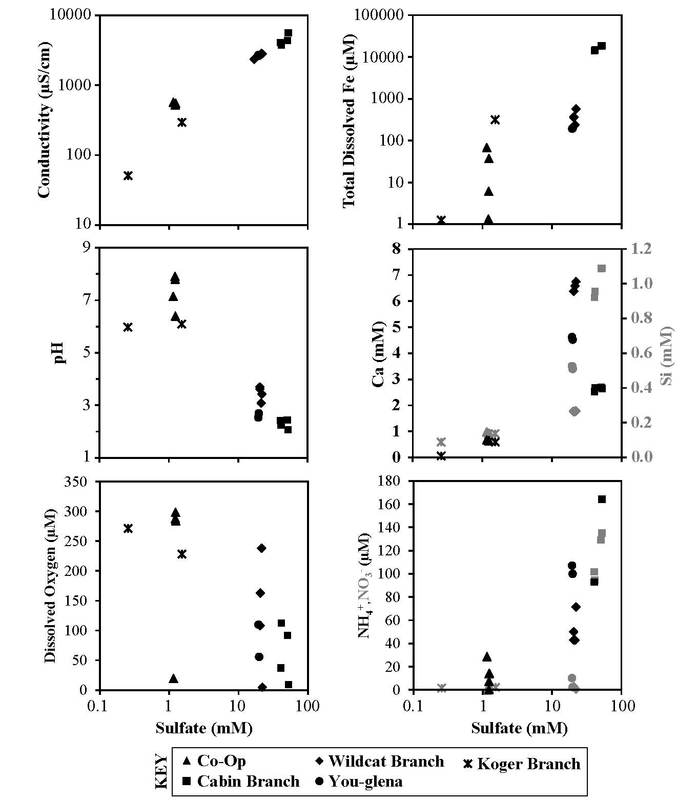
Havig et al. (2017) Figure 3: Comparison geochemistry for sampled acid mine drainage sites, with conductivity, pH, dissolved oxygen concentration, total dissolved iron concentration, calcium and silica concentration, and ammonium and nitrate concentration plotted against sulfate concentration. Concentrations given in either mmol/L (mM) or µmol/L (µM).
|
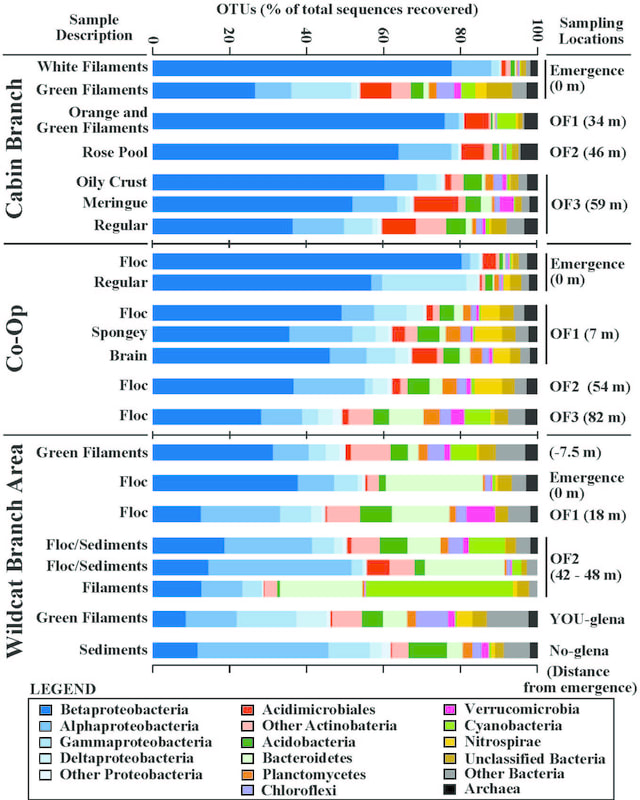
Havig et al. (2017) Figure 6: Microbial community compositions for the Cabin Branch sample sites, Co-Op Branch sample sites, and Wildcat Branch Area sample sites. Source = emergence, OF = outflow. X-axis gives biofilm descriptive names on the left and distances from emergence on the right.
|
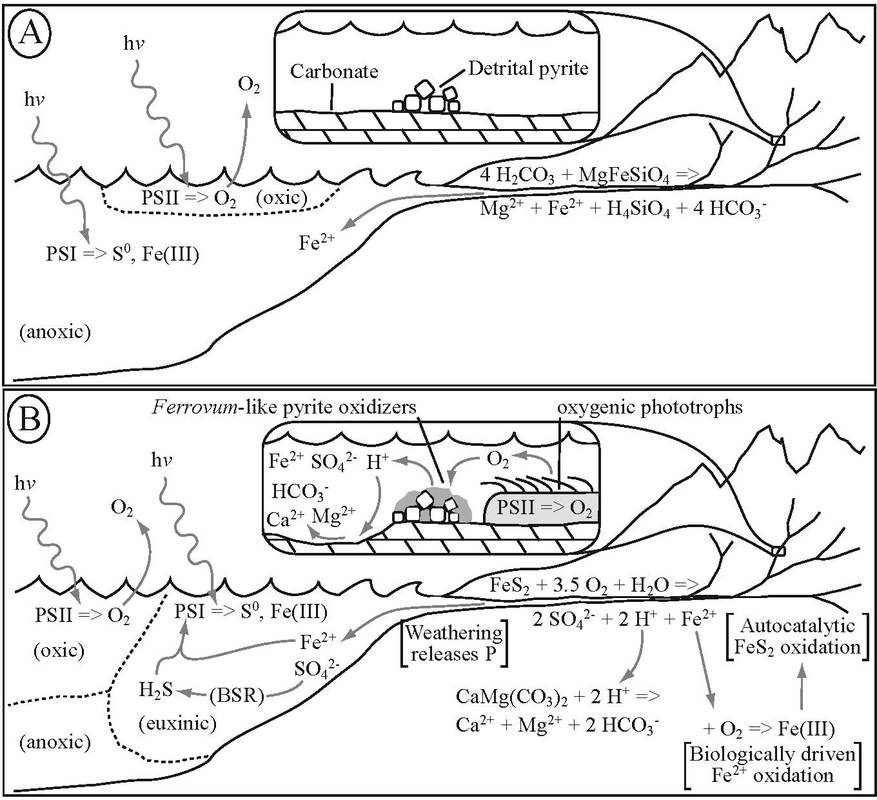
Havig et al. (2017) Figure 8: Conceptual model showing relationship between oxygen production by oxygenic photoautotrophs, AMD-type weathering driven by Ferrovum-like pyrite oxidizers, and euxinic conditions on continental surfaces and in the ocean for the A) Early Neoarchean through the B) Late Neoarchean to the Great Oxidation Event. PSI = photosystem I (anoxygenic photosynthesis), PSII = photosystem II (oxygenic photosynthesis), BSR = biological sulfate reduction.
|